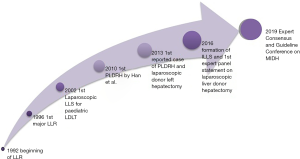
History of laparoscopic hepatectomy in donor
Laparoscopic Surgery has been established as a standard in the surgical specialty. It is adopted in most centers for major surgeries however it has been introduced to liver surgery fairly recently. Even though the first laparoscopic liver resection (LLR) was performed way back in 1992 it is still being used only in select centers with high volumes and by experienced surgeons. This is primarily due to technical difficulties faced in liver surgery.
History of laparoscopic liver surgery
So far 3 international consensus meetings on LLR have been held. The first International Consensus Conference on Laparoscopic Liver Resection (ICCLLR) was held in Louisville, KY, USA (2008). The practicality of performing LLR was discussed (1) with a special focus on safety. Laparoscopy was recommended for select cases with small tumors in segments 2–6 and left lateral sectionectomy (LLS). Its use in major liver resection was considered unfeasible. Nguyen et al reported 127 series of 3000 cases of LLR worldwide. The operations were considered safe with a morbidity of 10.5% and mortality of 0.3% (2). The second meeting of ICCLLR was held in Morioka, Japan in 2014. It recommended 3 actions for patients protection; prospective reporting registry, difficulty scoring system and creation of a formal education structure (3). It focused on comparisons with open technique and demonstrated benefits of laparoscopic approach (4). The International Laparoscopic Liver Society (ILLS) was officially launched at the International Hepato-pancreato-Biliary Association meeting in Sao Paulo in April 2016 (5).
The launch of minimally invasive donor hepatectomy (MIDH) in paediatric patients
Much before the ICCLLR, the first pure laparoscopic donor hepatectomy in pediatric transplantation was reported in France in 2002 (6). Laparoscopic LLS is a safe procedure as the anatomy of the lateral section is favorable with limited anatomical variations. It is possible to obtain grafts similar to the open technique (7). Laparoscopic LLS for pediatric living donor liver transplant (LDLT) has shown similar short-term outcomes as laparoscopic donor nephrectomy, suggesting that the laparoscopic approach should be considered the new standard practice (8).
Laparoscopic major hepatectomy in living donors
Laparoscopic surgery for major hepatic resections was being considered dangerous due to the complex anatomy of the liver. However Koffron et al. (9) performed the first laparoscopic assisted right hepatectomy in 2006 which was later followed by pure laparoscopic donor right hepatectomy (PLDRH) by Han et al. in 2010 but reported in 2015 (10). Since then, laparoscopic surgery for major hepatectomy started being adopted at many centers. A report of 9 donors undergoing either pure laparoscopic or hand assisted laparoscopic right hepatectomy safely, preserving branches of the middle hepatic vein was published in 2009 further proving the feasibility of the procedure. Another systematic review in Sao Paolo focused on outcomes of hybrid versus open and hybrid versus pure laparoscopic surgery. Twenty-one studies were included. The hybrid procedure has less blood loss, shorter hospital stay and lower morbidity compared to open. There was no difference when compared to the pure laparoscopic technique. They concluded that hybrid donor hepatectomy should be considered as a minimal invasive approach (11). Giulianotti et al. reported the first robotic assisted donor right hepatectomy in 2012 (12). The results of robotic hepatectomy were similar to that of PLDRH; operation time was longer but required less analgesics and had a quicker recovery compared to open surgery. The 3D vision allows depth perception and improves pure surgical performance (13). The first reported cases of PLDRH were in 2013 by Soubrane et al. (14) and Rotellar et al. (15) Laparoscopic donor hepatectomy aims to provide equal donor safety and faster rehabilitation compared to the open technique for living donors. The risk of mortality and morbidity depends on three factors: the physiological status, the proportion of liver excised, and, the amount of intraoperative blood loss and need for transfusion. Thus, to reduce morbidity in living liver donors, transplant teams must focus on the best technique of surgery in the donor, leaving adequate liver remnant with the lowest blood loss (14). Around the same time 2 case reports of pure laparoscopic donor left hepatectomy were published by Samstein et al. (16) and Troisi et al. (17).
The first expert panel statement on laparoscopic living donor hepatectomy was prepared during the third ICCLLR at the 26th World Congress of the International Association of Surgeons, Gastroenterologists and Oncologists (IASGO 2016) in Seoul (18). A 24 question survey was sent via email to 17 surgeons. The panel included surgeons who already perform laparoscopic donor hepatectomy, surgeons who perform LLR without laparoscopic donor hepatectomy, and surgeons who perform LDLT without laparoscopic surgery. The panel concluded that laparoscopic donor right hepatectomy is still innovative and in the developmental stage, and can only be recommended for performance by surgeons experienced in both LLR and LDLT. Some experts recommended that the donor indications for laparoscopic right hepatectomy should be a graft-to-recipient weight ratio >1.0, a remnant liver volume >35%, and a normal bile duct anatomy and portal vein (19) (Figure 1).
Data on the safety and feasibility of laparoscopic donor hepatectomy
In a study conducted including 72 donors in each group, the safety of PLDRH compared to open was assessed. Major complications were 15.4% in laparoscopic group and 6.6% in open group. However, after propensity score matching the results in both groups were similar. They also found that a right hepatic duct <1 cm on preoperative Magnetic Resonance Cholangiopancreatography was independently associated with complications in PLDRH (20). Another review of 100 initial cases of PLDRH compared its feasibility with open surgery. Their results showed longer operation time but lower blood loss in the laparoscopic group. Clavien dindo Grade III3b complications and above were higher in the laparoscopic group however the results were significant only in the first 25 cases. Grade IIIb complications were comparable when type 1 bile duct donors were selected. They thus concluded that laparoscopic living donor hepatectomy can cause significant complications initially hence donor selection must be carefully done (21). Similar results were shown in a center in Seoul where 115 donors were assessed. They reported higher grade IIIb complications in the initial period however no complications in the recent period. The difference in hemoglobin and AST in preoperative and postoperative period were also lower in the recent period. They considered performance of about 60 PLDRHs over 1 year to be sufficient to standardize the procedure (22).
In a 13-year experience study in France, when assessing cases of donor laparoscopic LLS, laparoscopic donor left hepatectomy and laparoscopic donor right hepatectomy, the authors concluded that the laparoscopic procedures require expertise in both open LDLT and LLR. The learning curve for LLR was considered to be 15–60 cases while mastery of open technique was considered mandatory. For beginners in laparoscopic LDLT, left lobe should be mastered first due to fewer vascular and biliary anatomical variations (23). A comparative study of laparoscopic versus open donor right hepatectomy after completion of learning curve showed no significant differences in terms of mean operative time, blood loss, graft weight, hospital stay thus showing PLDRH can be performed as safely as the open technique without increasing the risk of complications once sufficient experience has been obtained (24). A meta-Analysis in China evaluated 9 studies with 318 donors who underwent laparoscopic living donor hepatectomy. It demonstrated less post-operative blood loss, shorter hospital stay but longer operative time in the laparoscopic group. No significant differences were found in overall complications including bile leakage, postoperative bleeding, wound complications and time to dietary intake (25).
At the 24th annual congress of the International liver transplant society in 2018, laparoscopic donor hepatectomy in living donors was discussed. It was reported that results from centers routinely performing PLDRH indicate that donor complications and operative times decrease significantly with increasing experience, in particular when the learning curve (of around 60 cases) has been met. The use of advanced visualization techniques, such as 3-dimensional laparoscopy and indocyanine green (ICG) near-infrared fluorescence cholangiography, can also improve the safety of PLDRH in donors with complex portal vein anatomy. Biliary complications remain a significant concern with minimally invasive donor procedures (26). Laparoscopic MIDH is currently considered to be in the exploratory phase (IIb) under the Balliol classification scheme of the IDEAL model (27). It is now being widely performed at centers with the maximum cases being concentrated in Seoul, South Korea. This has established that pure laparoscopic MIDH can be performed extensively without damage to the donor and preserving graft outcomes. (Daniel Cherqui, Kyung-Suk Suh, Expert Consensus and Clinical Guidelines Conference in Minimally Invasive Donor Hepatectomy (MIDH), APHPBA, unpublished data).
There are no clear guidelines or recommendations to perform Laparoscopic MIDH hence in an attempt to develop guidelines an Expert Consensus and Guideline Conference on MIDH was held at the Asian-Pacific Hepato-Pancreato-Biliary Association meeting at Seoul on 7th September 2019. A team of experts after performing extensive research and reviewing existing literature formulated 18 questions covering Rationale of MIDH, Donor safety and outcomes, Recipient outcomes, technical issues and training and certification. The questions were voted upon by the surgeons present and the final guidelines are awaited.
Laparoscopic donor LLS is now considered standard practice. Adult laparoscopic donor liver transplant is recommended in high volume centers. The advances in imaging and technology, 3-dimensional surgical imaging, use of ICG fluorescence imaging have made laparoscopic major hepatectomy more widespread and safer than what was earlier assumed. Strict selection criteria, standardization of the procedure, supervised training under experienced mentors will help in reducing the learning curve and thus spread the use of LLR and later laparoscopic MIDH to more centers making it easily accessible to the donor.
Acknowledgments
Funding: None.
Footnote
Provenance and Peer Review: This article was commissioned by the Guest Editors (Kwang-Woong Lee and Jeong-Moo Lee) for the series “Pure Laparoscopic Donor Hepatectomy” published in Laparoscopic Surgery. The article has undergone external peer review.
Conflicts of Interest: All authors have completed the ICMJE uniform disclosure form (available at http://dx.doi.org/10.21037/ls.2019.12.05). The series “Pure Laparoscopic Donor Hepatectomy” was commissioned by the editorial office without any funding or sponsorship. The authors have no other conflicts of interest to declare.
Ethical Statement: The authors are accountable for all aspects of the manuscript in ensuring that questions related to the accuracy or integrity of any part of the manuscript are appropriately investigated and resolved.
Open Access Statement: This is an Open Access article distributed in accordance with the Creative Commons Attribution-NonCommercial-NoDerivs 4.0 International License (CC BY-NC-ND 4.0), which permits the non-commercial replication and distribution of the article with the strict proviso that no changes or edits are made and the original work is properly cited (including links to both the formal publication through the relevant DOI and the license). See: https://creativecommons.org/licenses/by-nc-nd/4.0/.
References
- Buell JF, Cherqui D, Geller DA, et al. Position on laparoscopic liver surgery. Ann Surg 2009;250:825-30. [Crossref] [PubMed]
- Nguyen KT, Gamblin TC, Geller DA. World review of laparoscopic liver resection-2,804 patients. Ann Surg 2009;250:831-41. [Crossref] [PubMed]
- Wakabayashi G, Cherqui D, Geller DA, et al. Recommendations for laparoscopic liver resection: a report from the second international consensus conference held in Morioka. Ann Surg 2015;261:619-29. [PubMed]
- Cho JY, Han HS, Wakabayashi G, et al. Practical guidelines for performing laparoscopic liver resection based on the second international laparoscopic liver consensus conference. Surg Oncol 2018;27:A5-A9. [Crossref] [PubMed]
- Cherqui D, Wakabayashi G, Geller DA, et al. The need for organization of laparoscopic liver resection. J Hepatobiliary Pancreat Sci 2016;23:665-7. [Crossref] [PubMed]
- Cherqui D, Soubrane O, Husson E, et al. Laparoscopic living donor hepatectomy for liver transplantation in children. Lancet 2002;359:392-6. [Crossref] [PubMed]
- Soubrane O, Cherqui D, Scatton O, et al. Laparoscopic left lateral sectionectomy in living donors: safety and reproducibility of the technique in a single center. Ann Surg 2006;244:815-20. [Crossref] [PubMed]
- Soubrane O, de Rougemont O, Kim KH, et al. Laparoscopic Living Donor Left Lateral Sectionectomy: A New Standard Practice for Donor Hepatectomy. Ann Surg 2015;262:757-61; discussion 761-3. [Crossref] [PubMed]
- Koffron AJ, Kung R, Baker T, et al. Laparoscopic-assisted right lobe donor hepatectomy. Am J Transplant 2006;6:2522-5. [Crossref] [PubMed]
- Han HS, Cho JY, Yoon YS, et al. Total laparoscopic living donor right hepatectomy. Surg Endosc 2015;29:184. [Crossref] [PubMed]
- Coelho FF, Bernardo WM, Kruger JAP, et al. Laparoscopy-assisted versus open and pure laparoscopic approach for liver resection and living donor hepatectomy: a systematic review and meta-analysis. HPB (Oxford) 2018;20:687-94. [Crossref] [PubMed]
- Giulianotti PC, Tzvetanov I, Jeon H, et al. Robot-assisted right lobe donor hepatectomy. Transpl Int 2012;25:e5-9. [Crossref] [PubMed]
- Kwon CHD, Choi GS, Joh JW. Laparoscopic right hepatectomy for living donor. Curr Opin Organ Transplant 2019;24:167-74. [Crossref] [PubMed]
- Soubrane O, Perdigao Cotta F, Scatton O. Pure laparoscopic right hepatectomy in a living donor. Am J Transplant 2013;13:2467-71. [Crossref] [PubMed]
- Rotellar F, Pardo F, Benito A, et al. Totally laparoscopic right-lobe hepatectomy for adult living donor liver transplantation: useful strategies to enhance safety. Am J Transplant 2013;13:3269-73. [Crossref] [PubMed]
- Samstein B, Cherqui D, Rotellar F, et al. Totally laparoscopic full left hepatectomy for living donor liver transplantation in adolescents and adults. Am J Transplant 2013;13:2462-6. [Crossref] [PubMed]
- Troisi RI, Wojcicki M, Tomassini F, et al. Pure laparoscopic full-left living donor hepatectomy for calculated small-for-size LDLT in adults: proof of concept. Am J Transplant 2013;13:2472-8. [Crossref] [PubMed]
- Cho JY, Han HS, Kaneko H, et al. Survey Results of the Expert Meeting on Laparoscopic Living Donor Hepatectomy and Literature Review. Dig Surg 2018;35:289-93. [Crossref] [PubMed]
- Han HS, Cho JY, Kaneko H, et al. Expert Panel Statement on Laparoscopic Living Donor Hepatectomy. Dig Surg 2018;35:284-8. [Crossref] [PubMed]
- Park J, Kwon DCH, Choi GS, et al. Safety and Risk Factors of Pure Laparoscopic Living Donor Right Hepatectomy: Comparison to Open Technique in Propensity Score-matched Analysis. Transplantation 2019;103:e308-16. [Crossref] [PubMed]
- Rhu J, Choi GS, Kim JM, et al. Feasibility of total laparoscopic living donor right hepatectomy compared with open surgery: comprehensive review of 100 cases of the initial stage. J Hepatobiliary Pancreat Sci 2019; [Epub ahead of print]. [PubMed]
- Lee KW, Hong SK, Suh KS, et al. One Hundred Fifteen Cases of Pure Laparoscopic Living Donor Right Hepatectomy at a Single Center. Transplantation 2018;102:1878-84. [Crossref] [PubMed]
- Brustia R, Komatsu S, Goumard C, et al. From the left to the right: 13-year experience in laparoscopic living donor liver transplantation. Updates Surg 2015;67:193-200. [Crossref] [PubMed]
- Lee B, Choi Y, Han HS, et al. Comparison of pure laparoscopic and open living donor right hepatectomy after a learning curve. Clin Transplant 2019;33:e13683 [Crossref] [PubMed]
- Xu J, Hu C, Cao HL, et al. Meta-Analysis of Laparoscopic versus Open Hepatectomy for Live Liver Donors. PLoS One 2016;11:e0165319 [Crossref] [PubMed]
- De Martin E, Hessheimer A, Chadha R, et al. Report of the 24th Annual Congress of the International Liver Transplantation Society. Transplantation 2019;103:465-9. [Crossref] [PubMed]
- McCulloch P, Altman DG, Campbell WB, et al. No surgical innovation without evaluation: the IDEAL recommendations. Lancet 2009;374:1105-12. [Crossref] [PubMed]
Cite this article as: D’Silva M, Cho JY, Han HS, Yoon YS, Choi YR, Lee JS, Lee B, Kim J. History of laparoscopic hepatectomy in donor. Laparosc Surg 2020;4:15.